INTRODUCTION
Over the past two decades, botulinum toxin (BoNT) has emerged as an important clinical tool in the management of a variety of medical and aesthetic conditions, including blepharospasm, cervical dystonia, chronic migraine (CM), and glabellar wrinkles. A common thread underlying all these conditions is that symptoms depend on the release of excessive neurotransmitters and/or neuropeptides from motor or sensory neurons and increased excretory drive.
BoNTs are the products of several clostridial bacterial strains, most notably Clostridium botulinum. The neurotoxin produced by these bacteria specifically disrupts the normal release of vesicular neurochemicals, based on motor and sensory neuronal response in autonomic and somatic nervous systems. Such a disruptive mechanism explains the clinical utility of BoNT in such a broad range of indications.
Sleep bruxism (SB), a stereotyped oromandibular activity during sleep, is characterized by grinding and clenching of teeth. The prevalence of SB decreases from 14%–20% in childhood to 3% in old age and sex differences in SB prevalence are not recognized [1].
There are a variety of treatments for SB management, including occlusal instruments, behavioral approaches, and pharmacological management. However, the comprehensive management of SB has yet to be reported [2].
The most recent pharmacological approach for managing the pathological effects of nocturnal and diurnal bruxism entails the injection of BoNTs into the masticatory muscle. Recent studies show that BoNT is effective in controlling involuntary oral and facial movements and secondary bruxism in patients with movement disorders [3-6]. The masseter and temporal muscles (TMs) are known as SB-related muscles [7]. Many case studies have reported injections into the masseter muscle, while others involved injection into both masseter and TMs [8-10].
CM refers to migraine attacks occurring 15 days or more monthly for at least 3 months, with attacks lasting 4 hours or more. The Allergan protocol that was approved by the United States Food and Drug Administration involves the injection of 155 U of BoNT in a stereotypical pattern over the head and neck muscles [11,12]. The spread of the neurotoxin over a large area ensures that the neurotoxin not only affects the head and neck muscles that contribute to the sensitization but also centrally to modulate the release of glutamate, substance P and other neurotransmitters that contribute to the sensitization [13].
The use of BoNT for preventive migraine treatment has been well studied. Data from the Phase III Research Evaluating Migraine Prophylaxis Therapy (PREEMPT) trial showed a 50% reduction in headache frequency at all time points compared with placebo, and patients reported an improvement in health-related quality of life [11,12].
In this review, we compare the anatomical considerations for BoNT injection into TMs in 2 different diagnoses: SB and CM.
BoNT INJECTION INTO TMs IN SB
Many case reports have reported that BoNT injections are effective in controlling involuntary orofacial movements and secondary SB in patients with movement disorders, and awake bruxism (e.g., clenching habit). Although many studies are related to the use of BoNT to treat bruxism, few studies have described the BoNT injection protocol including drug dosage, dilution method, and injection site in the masticatory muscle. The previous studies were not comprehensive or accurate, nor did they explain the rationale for injection of BoNT into a specific site.
In a cadaver study, Choi et al. [14] proposed that BoNT should be injected into the temporalis muscle at least 45 mm vertically above the zygomatic arch to ensure that the muscle is targeted. However, this study focuses on the muscles, tendons and deep temporal nerve distributions.
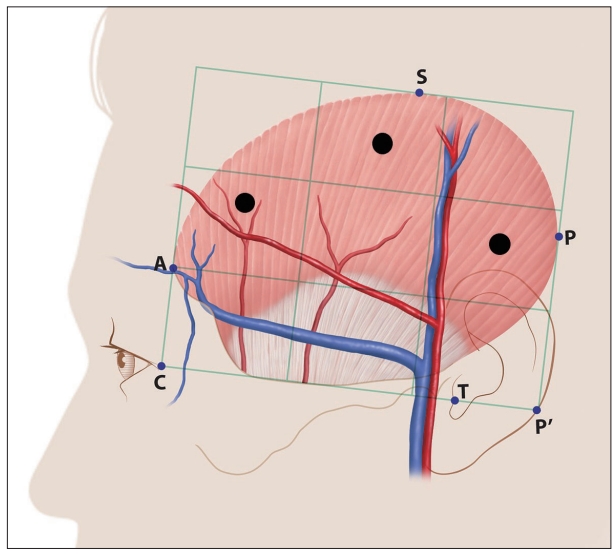
Lee et al. [15] reported a study simplifying the optimal temporal areas for safe and reproducible BoNT injections into the TM via detailed dissection and measurements of the structures in the temporal area, and to provide a virtual topographic map of postural relations involving the major anatomical structures, such as the superficial temporal artery, the middle temporal vein, and the temporal branch of the facial nerve in the TM.
The relative ratios of the anatomical location of the structures were constant in all the specimens. The reference line was first established as Canthion–Tragus (C-T), and the distance between C and E was set as the base of the TM rectangle. The vertical sides of the rectangle were configured at 80% of the base (ratio 5:4). It is convenient to additionally specify the exact location of the injection site within the nine TM compartments described here to enhance the clinician’s confidence to perform the injection (Fig. 1).
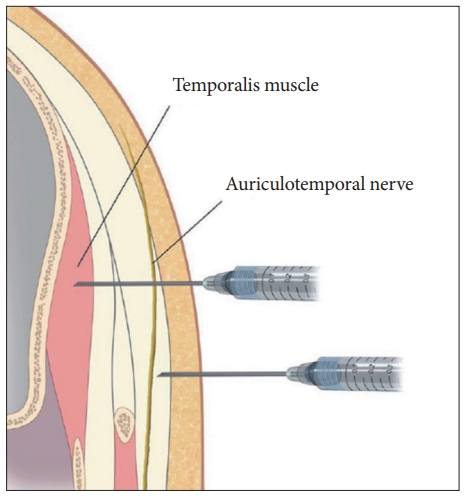
A 12.7-mm-long needle is generally recommended based on the thickness of the soft tissues. The needle initially touches the bone and the clinician withdraws the needle by 2–3 mm, followed by injecting a portion of the BoNT dose (Fig. 2).
BoNT INJECTION INTO TMs IN CM
BoNT injections to treat CM have evolved technologically to cover clear injection points in the head and neck area. Previous studies have suggested that BoNT should be injected intramuscularly into seven muscles in the head and neck.
According to the Allergan trainee workbook, the procedure should be started with the left temporalis muscle via fixed-dose injections, followed by any follow-the-pain injections, and the procedure repeated symmetrically on the right side as explained below.
Injection 1
- Ask the model to clench his or her teeth. Locate and palpate the anterior aspect of the temporalis muscle.
- Make the first injection approximately at two fingerbreadths behind this point.
- Try to stay behind the hairline.
Injection 2
Make the second injection –0.5 cm superior and 1 fingerbreadth (–1.5 cm) posterior to the first injection in the medial aspect of the muscle.
Injection 4
This should be –1.5 cm below and perpendicular to the second injection into the medial aspect of the muscle.
However, inconsistencies arise as the above protocol uses surface structures, such as hairlines, as landmarks. The proposed mechanism of analgesia suggests that the injection should be administered into the sensory nerve, and not the muscle. Optimal intervention can be performed via intensive observation of innervation of the target area based on consistent anatomical landmarks.
Recently, Kim et al. [16] proposed that the optimal BoNT-A injection point for CM is located in the temporal region. BoNT-A is injected superficially into the temporal region of the auriculotemporal nerve (ATN) distribution guided by clear and consistent anatomical landmarks and reference axes.
The first point is about 2 cm anterior and 3 cm superior to the two orthogonal reference lines based on the canthus and tragus, while the second point is located about 4 cm superior to the first point, and the third and fourth points are both approximately 2 cm superior to the first point, but 1 cm anterior and posterior, respectively. The agent needs to be injected because the ATN travels superficially over the temporalis muscle [17].
The ATN appears superficially in the anterosuperior region of the tragus, where the nerve is situated deeper than the superficial temporal artery and passes between the artery and the superficial temporal vein. The superficial ATN splits into anterior and posterior divisions. The posterior division projects in front of the ear and branches off into several segments supplying the skin, while the anterior division traverses in a superior direction. The area in which the distribution of the nerve was the greatest is presented in Fig. 3. BoNT must be injected subcutaneously because the ATN traverses superficially over the TM (Fig. 2).
CONCLUSION
Based on the anatomical considerations of BoNT injection into TMs, two different diagnoses including SB and CM are reviewed.
In patients with SB, the target area should be deep temporal nerves in TMs to ensure deeper injection into the muscles peripherally. Patients with CM should be superficially injected into the temporal region of the ATN distribution area under guidelines based on clearly identifiable and consistent anatomical landmarks and reference axes.