AbstractBackground and ObjectiveIndividuals diagnosed with obstructive sleep apnea (OSA) experience recurrent episodes of breathing cessation due to blockage of the upper airway during sleep. This study aimed to assess the Pediatric Sleep Questionnaire (PSQ) scores, Epworth Sleepiness Scale (ESS) scores, and Mallampati score in pediatric patients with OSA, compared with non-OSA.
MethodsAn observational study was conducted on 100 pediatric female patients aged 9-13 years with retrognathic mandible. The participants were subjected to PSQ, ESS, and Mallampati score to assess the size and shape of the patient’s tongue and esophageal airway. Finally, polysomnographic examination was carried out for all the participants and they were divided into two groups: Group 1 (50 patients) who were diagnosed with OSA by polysomnography and Group 2 (50 patients) consisting of non-OSA patients. Chi-square and Mann–Whitney U test were applied to compare scores between the two groups.
ResultsThe mean PSQ, ESS, and Mallampati scores of participants were 0.78 ± 0.45, 9.86 ± 2.47, and 2.27 ± 0.98, respectively in Group 1 and 0.24 ± 0.03, 7.27 ± 2.39, and 1.32 ± 0.57, respectively in Group 2, and this difference was statistically significant (p < 0.05). PSQ, ESS, and Mallampati scores had specificity of 100%, 95.5%, and 72.7%, respectively, whereas the sensitivity was 45.5%, 22.7%, and 63.6%, respectively. ESS scores ≥ 10, PSQ scores ≥ 0.3, and Mallampati scores ≥ 2 were indicative of OSA.
INTRODUCTIONPatients with obstructive sleep apnea (OSA) experience recurrent pause during sleep. This phenomenon is ascribed to blockage of the upper respiratory tract during sleep, which can be attributed to insufficient motor tone in the tongue or muscles responsible for dilatation of the airway. Apnea can be divided into three categories: central apnea, which involves a depressed respiratory center with no efferent output; obstructive apnea, which is characterized by blocked airflow leading to inadequate ventilation; and mixed apnea, which includes both central and obstructive components [1]. Decreased ventilation during sleep in children with increased body weight is associated with snoring, hypercapnia, and arousals during sleep [2]. The manifestation of pediatric obstructive sleep apnea has been observed to impact the attention span of children, potentially resulting in hyperactivity, decreased performance, and implications for the growth trajectory of affected individuals [3].
The diagnosis of OSA in children involves multiple stages. The symptoms exhibited by children are characterized by a higher level of complexity, necessitating a more comprehensive clinical evaluation. Relying only on basic assessment may lead to incorrect conclusions. To ascertain the medical background of individuals with suspected OSA, physicians employed questionnaires filled out by the parents or guardians of the affected children. The Pediatric Sleep Questionnaire (PSQ) is widely used as the primary instrument for detecting pediatric OSA. This questionnaire consists of 10 items that offer respondents fixed response alternatives [4]. Based on the current guidelines provided by the European Respiratory Society Task Force, the PSQ is regarded as a reliable instrument for identifying children affected by OSA, particularly those exhibiting an apnea-hypopnea index (AHI) beyond 5 [5].
The Epworth Sleepiness Scale (ESS) is a standardized tool developed to assess sleep propensity in a straight forward manner. The scale encompasses a comprehensive spectrum of sleep tendencies ranging from the most elevated to the most diminished. The ESS concept is derived from empirical observations pertaining to the characteristics and prevalence of diurnal sleep and somnolence. Certain individuals with extreme daytime sleepiness actively engage in various activities and deliberately refrain from assuming a reclined or relaxed position during the day, thereby consciously avoiding sleep. Individuals who experience boredom, possess surplus leisure time, or exhibit social withdrawal tendencies do not necessarily exhibit signs of drowsiness, opt to recline, or engage in daytime sleep [6]. Boari et al. [7] compared “ESS scores” and “AHI” and concluded that ESS can detect normal and severe levels of apnea.
Following its inception in the 1980s, the Mallampati score system expeditiously gained prominence as a customary preoperative physical examination technique employed to predict the likelihood of encountering a challenging airway [8]. There is a documented association between the Mallampati scores and OSA. OSA is a common sleep-related respiratory disease. Relaxation of upper airway structures during sleep results in blockage of the upper airway. The Mallampati score has been demonstrated to be an autonomous risk factor for predicting the existence and intensity of OSA. There is a positive correlation between an increase in the Mallampati score by one point and doubling of the odds of experiencing OSA. There is a positive correlation between the Mallampati score and the AHI, whereby a one-point increase in the former is associated with an average increase of more than five episodes per hour in the latter. Hence, the evaluation of a patient’s Mallampati score prior to polysomnography has potential utility in the clinical context for predicting OSA [9].
The literature search revealed that there was no holistic literature wherein the three different techniques were used to evaluate and compare OSA and non-OSA patients. Therefore, this study was conducted with the null hypothesis that there was no significant difference in the PSQ, ESS, and Mallampati scores between participants with OSA and those without OSA.
METHODSStudy DesignThis prospective study was conducted in the Department of Orthodontics after obtaining ethical clearance from the D Y Patil Dental College Institutional Review Board (594(5)/2022) and in accordance with the Declaration of Helsinki. Written informed consent from the parents and assent from the children were obtained following an explanation of the study, while ensuring confidentiality.
Sample Size CalculationThe sample size was calculated using G*Power software (Version 3.1.9; Heinrich-Heine-Universität Düsseldorf, Düsseldorf, Germany). The power analysis revealed that 90 samples would provide a power of 80%, with a type 1 error of 5% and a type 2 error of 20%. Considering a 10% dropout rate, the present study was conducted with 100 participants.
Eligibility CriteriaThe study period spanned from January 2022 to August 2023. Participants were recruited from the hospital outpatient department after they met the following inclusion and exclusion criteria: growing female patients in the age group of 9–13 years at cervical vertebrae maturation of stage 1 and 2, having skeletal class II due to retrognathic mandible (maxilla-mandibular relationship as assessed by angle between point A which is the deepest point on anterior concavity of maxillae and point B which is the deepest point on anterior concavity of the mandible as designated by ANB angle > 4°), narrow and constricted maxillary arch. Patients with a history of systemic disease, a history of previous orthodontic treatment, and those who were not willing to participate in the study were excluded. Participants were recruited to the study sample using the purposive sampling technique. Patients were then assessed for weight, height, and neck circumference. The height, age, sex, and weight of the patients were entered into the calculator for body mass index (BMI) computation. The BMI percentile of the participants was calculated based on the BMI values. BMI Z-scores were also calculated. The Center for Disease Control and Prevention defined obesity as at or above the 95th percentile of BMI for age and overweight as between the 85th to 95th percentile of BMI for age, and normal weight as between the 5th and 85th percentile of BMI [10].
Study MethodologyA total of 495 participants were screened according to the eligibility criteria and 160 were finally selected. A total of 160 participants were subjected to a PSQ, ESS, and Mallampati scoring. The PSQ consists of 22 questions that inquire about snoring frequency, loud snoring, observed apnea, difficulty breathing while sleeping, daytime sleepiness, inattentive or hyperactive behavior, and other characteristics of pediatric OSA [4]. The responses are classified using a coding system where “yes” is represented by the number 1, “no” is represented by the number 0, and “don’t know” is represented as missing. The PSQ score is determined by calculating the ratio between the sum of “yes” responses and the number of questions that were answered. A PSQ score equal to or greater than 0.33 is considered to be indicative of OSA [4].
The ESS is a self-administered questionnaire that consists of eight questions. Participants were asked to assess their likelihood of falling asleep or dozing off during eight different activities, using a four-point scale (ranging from 0 to 3). The majority of individuals engage in these activities sporadically, not daily. The ESS score, which is the summation of scores ranging from 0 to 3, exhibits a potential range of values from 0 to 24. A score 0 means never dozing, 1 means a slight chances of dozing, 2 means a moderate chance of dozing, and 3 indicates a high chance of dozing. ESS scores can be interpreted as follows: 0–10, normal daytime sleepiness; 11–12, mild excessive daytime sleepiness (EDS); 13–15, moderate EDS; and 16–24, severe EDS. The average sleep propensity in an individual’s everyday life, also known as “daytime sleepiness,” has an inverse relationship with the ESS score. EDS was accepted in individuals with a total score of 10 or higher [6].
Mallampati scoring is a useful tool for assessing the likelihood of OSA in an individual, with scores based on a scale of 1 to 4, which assesses the size and shape of a patient’s tongue and esophageal airway. The patients were permitted to produce sounds while their mouths were in an open position. Scoring is contingent on the discernibility of anatomical formations within the oropharynx. Score 1–4 were given based on the classes as follows: Class 1 provided visibility of the tonsils, uvula, and both the soft and hard palates. Class 2 allowed for the visibility of the soft palate and at least a portion of the uvula. Class 3 granted visibility to the soft palate. Finally, class 4 did not provide visibility beyond the tongue, encompassed the faucial/tonsillar pillars (arches positioned anteriorly and posteriorly to the tonsils), foundation of the uvula, and soft palate. Notably, the magnitude of the tonsils did not affect the Mallampati score [8].
After completion of the PSQ, ESS, and Mallampati scoring, all patients underwent polysomnographic examination. A level 1 testing protocol was employed for polysomnographic examination (Alice 5 Philips Respironics PSG; MFI Medical Equipment Inc., San Diego, CA, USA), necessitating overnight sleep in a sleep laboratory under the supervision of a trained technician. This procedure entailed recording a minimum of seven data channels (although typically ≥16), encompassing respiratory, cardiovascular, and neurologic parameters, to generate an allencompassing representation of sleep architecture. Polysomnography records mental waves, blood oxygen level, pulse, and breathing during rest. An AHI score less than 5 were categorized as non-OSA patients [11]. The current study employed AHI standards that were applicable to adults, rather than children. Previous studies have revealed that there is no discernible difference in the consequences between a child with an AHI less than 1 event per hour and one with an AHI ranging from 1 to 5 events per hour [12]. The study that provided a classification system for paediatric OSA [13], based on the AHI index, was restricted by a limited sample size, the absence of electroencephalography validation of sleep, and the disregard of hypopneas or central events. The determination of normative standards for PSG was based on the statistical distribution of data and it has not been established that these standards hold any validity as predictors of long-term outcomes [14]. Fifty cases of OSA were diagnosed in 160 cases, which formed Group 1. To follow a 1:1 allocation ratio and according to sample size calculation, 50 non-OSA cases were randomly selected from the 110 non-OSA cases for analysis (Group 2). The study design is shown Fig. 1.
Statistical AnalysisData were evaluated for completeness and transferred to Microsoft Excel Version 13 for statistical analysis. Statistical analysis was performed at a 95% confidence level with a statistical significance of p-value less than 0.05. Statistical analysis was performed using IBM SPSS Statistics (Ver 23; IBM Corp., Armonk, NY, USA). For categorical variables, the frequency and percentage for the data were obtained, whereas for continuous variables, the mean and standard deviation for the data were obtained. Shapiro–Wilk test was used to assess the normality of data and as data was not normally distributed; therefore, the non-parametric tests such as chi-square and Mann–Whitney U test were applied to compare the scores between the two groups. Furthermore, the sensitivity and specificity of PSQ, ESS, and Mallampati score was evaluated using receiver operating characteristic (ROC) curves, and cut-off values were calculated using Youden index.
RESULTSThe null hypothesis was rejected in the present study as significant differences were observed between the groups. The mean age of the patients in Group 1 was 11.9 ± 2.1 years, and 11.3 ± 1.9 years in Group 2. The number of obese participants in the OSA and non-OSA groups was 12 and 10, respectively, whereas the number of participants with a normal BMI percentile was 20 in the OSA group and 29 in the non-OSA group. The mean BMI of patients with OSA was 34.1 ± 5.9 while it was 22.3 ± 8.5 in non-OSA group. The mean neck circumference in OSA patients was 18.62 ± 3.81 cm while in non-OSA patients was 12.15 ± 1.01 cm, respectively (Table 1). Most of the participants in Group 1 had mild (44%) and moderate (36%) ESS scores, whereas in Group 2, most of the participants had normal (56%) and mild (36%) ESS scores (Table 2). Most of the participants in Group 1 had class 3 (44%) and class 4 (24%) scores indicative of OSA, whereas in Group 2, most of the participants had class 1 (64%) and class 2 (32%) scores (Table 3).
The mean PSQ scores of the OSA group was 0.78 ± 0.45 and in the non-OSA group was 0.24 ± 0.03. This difference was statistically significant (p < 0.05). Similarly, statistically significant differences were observed between the groups for ESS and Mallampati scores, with Group 1 having higher scores than Group 2 (p < 0.05), as shown in Table 4.
ROC curves for PSQ, ESS, and Mallampati scores revealed that PSQ had 100% specificity, sensitivity of 45.5%, accuracy of 72.7%, and p-value of 0.997, whereas ESS had 95.5% specificity, sensitivity of 22.7%, accuracy of 59.1%, and p-value of 0.03. Mallampati score had 72.7% specificity, 63.6% sensitivity, accuracy of 68.2%, and p-value of 0.002. Mallampati had acceptable accuracy to diagnose OSA (area under curve [AUC] of 0.754), whereas ESS and PSQ had excellent accuracy in diagnosing OSA (AUC of 0.794 and 0.812, respectively). ESS scores above 10, PSQ scores above 0.3, and Mallampati scores more than or equal to 2 were indicative of OSA (Fig. 2).
DISCUSSIONOSA is characterized by specific features including recurrent constriction or obstruction of the upper airway during sleep, which causes frequent decreases in oxygen levels and subsequent arousal. Neglecting to manage may have detrimental impacts on an individual’s neurocognitive capabilities. Studies conducted by Talla et al. [15] and Triplett et al. [16] demonstrated a comparatively greater incidence of OSA individuals presenting with skeletal class II malocclusions, than in those with skeletal class I malocclusions.
This study aimed to compare the PSQ, ESS, and Mallampati scores of skeletal class II children with and without OSA. Ramar et al. [17] stated that the diagnosis and continuous care of OSA and primary snoring are normally responsible for sleep physicians, as outlined in the clinical practice guidelines issued by American Academy of Sleep Medicine and the American Academy of Dental Sleep Medicine.
The sleep quality scores of patients with and without OSA were compared using different tools. In the OSA group, the participants had an average PSQ score of 0.78 with a standard deviation of 0.45. In contrast, the non-OSA group had an average PSQ score of 0.24 with a standard deviation of 0.03 indicating good-quality sleep. In the OSA group, according to the Mallampati score examination, most patients had class 2 and 3, whereas most patients in the non-OSA group had class 1.
In previous studies using the Mallampati and ESS scores to assess the sleep quality of pediatric patients with OSA, it was found that poor quality scores indicate poor sleep quality [18-20]. The results of the present study are consistently in the line with the literature that was published universally [21-23]. On average, the odds of having OSA increased by more than 6-fold for every point increase in the Mallampati score [18]. This approach can result in earlier diagnosis and timely management of OSA. A delayed diagnosis of OSA not only leads to a diminished quality of life and various neurological and cardiopulmonary complications but also increases the financial burden on healthcare systems. Polysomnography is considered the gold standard for diagnosing pediatric OSA, and the Mallampati score serves as an additional resource to aid in the identification of patients with a heightened risk of pediatric OSA [21]. Despite numerous debates, studies have suggested that apnea/hypoxemia provides a substantial explanation for EDS in patients with OSA [22,23].
When ESS, PSQ, and Mallampati were studied as reliable indicators of OSA by ROC curves, it was noticed that the highest specificity, accuracy, and AUC was noticed for PSQ, followed by ESS and Mallampati. The cut-off values for PSQ, ESS, and Mallampati were in accordance with previous studies [9,24,25]. Based on the findings of the present study, it can be concluded that PSQ and ESS had lower sensitivity, and Mallampati scoring was relatively reliable with satisfactory specificity and sensitivity (72.7%, and 63.6%, respectively). The reason for low sensitivity of PSQ and ESS might be due to fact that sensitivity of a questionnaire can be influenced by various factors such as social, cultural, and developmental differences. Questionnaires may not cover the full range of pediatric sleep disorders or may not be sensitive to specific conditions that are more prevalent in children including issues like night terrors, bedwetting, or parasomnias, which may not be fully addressed by these questionnaires. Moreover, both questionnaires rely on self-reporting or reporting by parents/guardians. Subjective measures can be influenced by individual perceptions, and children or their caregivers may not always accurately report sleep-related issues. The key strength of the study was a complete assessment of the patients with regards to PSQ, ESS, and Mallampati scores among participants with OSA and those without OSA.
LimitationsThis study was a single center study, involving female patients. The present study used AHI index of adults, and therefore, could have affected the grouping of patients into OSA and non-OSA groups. Hence, future multicenter studies are required to be conducted on both the genders with a large sample size, using AHI standards of children.
ConclusionPSQ, ESS, and Mallampati scores were higher among participants with OSA. PSQ and ESS had lower sensitivity (45.5% and 22.7%, respectively) and therefore, are unsuitable for diagnosing true positive cases of OSA. However, due to high specificity and accuracy of PSQ and ESS, they can be used for initial screening of OSA cases. Amongst all the methods, Mallampati scoring had a specificity of 72.7% and sensitivity of 63.6%, and therefore can be used satisfactorily in diagnosing OSA cases.
NOTESAvailability of Data and Material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author Contributions
Conceptualization: Chaitra Santoshkumar Mastud, Sonali V. Deshmukh, Jayesh Rahalkar. Data curation: Chaitra Santoshkumar Mastud. Formal analysis: Chaitra Santoshkumar Mastud, Madhusudhan Bharatwal, Shailaja Mane, Santoshkumar Pandurang Mastud. Investigation: Chaitra Santoshkumar Mastud, Madhusudhan Bharatwal, Shailaja Mane, Santoshkumar Pandurang Mastud. Methodology: Chaitra Santoshkumar Mastud, Sonali V. Deshmukh, Jayesh Rahalkar. Project administration: Chaitra Santoshkumar Mastud, Madhusudhan Bharatwal, Shailaja Mane, Santoshkumar Pandurang Mastud. Resources: Chaitra Santoshkumar Mastud, Sonali V. Deshmukh, Jayesh Rahalkar. Software: Santoshkumar Pandurang Mastud. Supervision: Chaitra Santoshkumar Mastud. Validation: Chaitra Santoshkumar Mastud, Shailaja Mane, Madhusudhan Bharatwal. Visualization: Chaitra Santoshkumar Mastud, Sonali V. Deshmukh, Jayesh Rahalkar. Writing—original draft: Chaitra Santoshkumar Mastud. Writing—review & editing: all authors.
REFERENCES1. Kondamudi NP, Khetarpal S. Apnea in children. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2023 [accessed 2023 August 23]. Available from: https://www.ncbi.nlm.nih.gov/books/NBK441894/.
2. Fiorino EK, Brooks LJ. Obesity and respiratory diseases in childhood. Clin Chest Med 2009;30:601-8.
3. Benninger M, Walner D. Obstructive sleep-disordered breathing in children. Clin Cornerstone 2007;9(Suppl 1):S6-12.
4. Chervin RD, Hedger K, Dillon JE, Pituch KJ. Pediatric sleep questionnaire (PSQ): validity and reliability of scales for sleep-disordered breathing, snoring, sleepiness, and behavioral problems. Sleep Med 2000;1:21-32.
5. Kaditis AG, Alonso Alvarez ML, Boudewyns A, Alexopoulos EI, Ersu R, Joosten K, et al. Obstructive sleep disordered breathing in 2- to 18-year-old children: diagnosis and management. Eur Respir J 2016;47:69-94.
6. Johns MW. A new method for measuring daytime sleepiness: the Epworth Sleepiness Scale. Sleep 1991;14:540-5.
7. Boari L, Cavalcanti CM, Bannwart SR, Sofia OB, Dolci JEL. Evaluation of Epworth Sleepiness Scale in patients with obstructive sleep apneahypopnea syndrome. Rev Bras Otorrinolaringol 2004;70:752-6.
8. Mallampati SR, Gatt SP, Gugino LD, Desai SP, Waraksa B, Freiberger D, et al. A clinical sign to predict difficult tracheal intubation: a prospective study. Can Anaesth Soc J 1985;32:429-34.
9. Nuckton TJ, Glidden DV, Browner WS, Claman DM. Physical examination: Mallampati score as an independent predictor of obstructive sleep apnea. Sleep 2006;29:903-8.
10. Flegal KM, Wei R, Ogden C. Weight-for-stature compared with body mass index-for-age growth charts for the United States from the Centers for Disease Control and Prevention. Am J Clin Nutr 2002;75:761-6.
11. American Academy of Sleep Medicine Task Force. Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. Sleep 1999;22:667-89.
12. Goldstein NA, Sculerati N, Walsleben JA, Bhatia N, Friedman DM, Rapoport DM. Clinical diagnosis of pediatric obstructive sleep apnea validated by polysomnography. Otolaryngol Head Neck Surg 1994;111:611-7.
13. Section on Pediatric Pulmonology, Subcommittee on Obstructive Sleep Apnea Syndrome. Clinical practice guideline: diagnosis and management of childhood obstructive sleep apnea syndrome. Pediatrics 2002;109:704-12.
14. Brooks DM, Brooks LJ. Reevaluating norms for childhood obstructive sleep apnea. J Clin Sleep Med 2019;15:1557-8.
15. Talla R, Kamble R, Mathew JA, Nambiar K. Estimation of incidence and awareness of obstructive sleep apnea in class II malocclusion in young age adult population as compared to class I malocclusion in Vidharba population using questionnaire and lateral cephalogram. J Res Med Dent Sci 2022;10:6-12.
16. Triplett WW, Lund BA, Westbrook PR, Olsen KD. Obstructive sleep apnea syndrome in patients with class II malocclusion. Mayo Clin Proc 1989;64:644-52.
17. Ramar K, Dort LC, Katz SG, Lettieri CJ, Harrod CG, Thomas SM, et al. Clinical practice guideline for the treatment of obstructive sleep apnea and snoring with oral appliance therapy: an update for 2015. J Clin Sleep Med 2015;11:773-827.
18. Kumar HV, Schroeder JW, Gang Z, Sheldon SH. Mallampati score and pediatric obstructive sleep apnea. J Clin Sleep Med 2014;10:985-90.
19. Ali NJ, Pitson DJ, Stradling JR. Snoring, sleep disturbance, and behaviour in 4-5 year olds. Arch Dis Child 1993;68:360-6.
20. Gislason T, Benediktsdóttir B. Snoring, apneic episodes, and nocturnal hypoxemia among children 6 months to 6 years old. An epidemiologic study of lower limit of prevalence. Chest 1995;107:963-6.
21. Zreaqat M, Hassan R, Samsudin AR, Stas Y, Hanoun A. Tonsil size and Mallampati score as clinical predictive factors for obstructive sleep apnea severity in children. J Contemp Dent Pract 2021;22:850-3.
22. Lee SJ, Kang HW, Lee LH. The relationship between the Epworth Sleepiness Scale and polysomnographic parameters in obstructive sleep apnea patients. Eur Arch Otorhinolaryngol 2012;269:1143-7.
23. Basak H, Anadolu RY. Correlation between self-reported and questioned Epworth Sleepiness Scale results including polysomographic outcomes. Egypt J Otolaryngol 2022;38:109.
Fig. 2.Receiver operating characteristic curve for different scoring methods in study group: ESS (A), PSQ (B), and Mallampati score (C). ESS, Epworth Sleepiness Scale; PSQ, Pediatric Sleep Questionnaire; AUC, area under curve; CI, confidence interval. 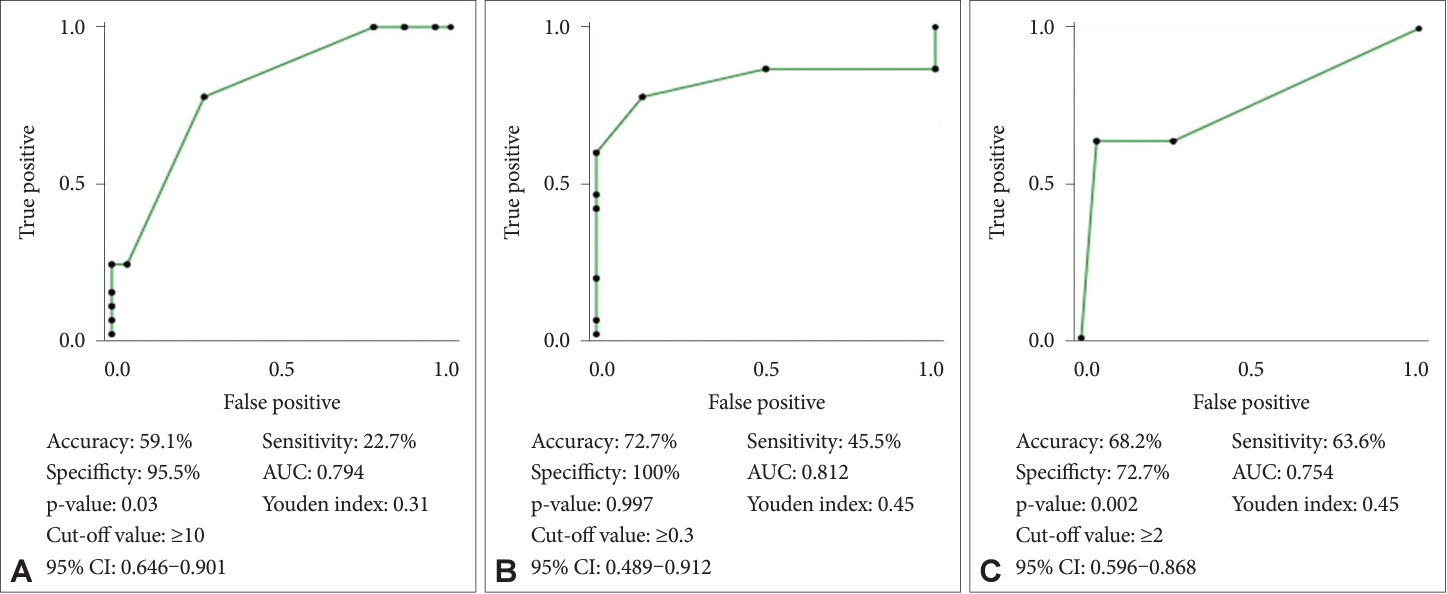
Table 1.Descriptive analysis of primary variables in study groups
Table 2.Frequency distribution of participants according to Epworth Sleepiness Scale
Table 3.Frequency distribution of participants according to Mallampati scores
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||