INTRODUCTION
Obstructive sleep apnea (OSA) is a respiratory disorder that occurs during sleep [1]. Recurrent pharyngeal airway collapse during respiration and the subsequent episodes of apnea, hypopnea, and arousal from sleep adversely impact the patientŌĆÖs quality of life and elevate the risk of neurocognitive, pulmonary, and cardiovascular diseases through autonomic dysregulation [2].
Therefore, it is essential for individuals experiencing OSA symptoms to undergo medical evaluation and receive appropriate treatment. Therapeutic options include lifestyle modifications such as weight loss, positional therapy, positive airway pressure (PAP), oral appliance therapy, and surgical modifications of the upper airway. Treatment is typically determined by the patientŌĆÖs physical examination findings, results from sleep tests, and the patientŌĆÖs opinion.
While PAP remains the mainstay treatment of OSA, oral appliance therapy is proposed as an alternative therapeutic option for patients who show intolerance for PAP [3]. Oral appliances reduce apnea/hypopnea by expanding the pharyngeal airway via mandible and tongue advancement [4,5]. In addition, various studies are conducted to achieve better individualized treatment outcomes through patient selection and device design as well as to compare the effects between the oral appliances and conventional PAP.
This review provides a comprehensive description of the various aspects of oral appliances used in the treatment of OSA, including their characteristics, clinical benefits, and possible side effects.
TYPES OF ORAL APPLIANCE
Mainly, oral appliances are categorized into three types: mandibular advancement devices (MADs), tongue-retaining devices (TRDs), and palatal lift appliances.
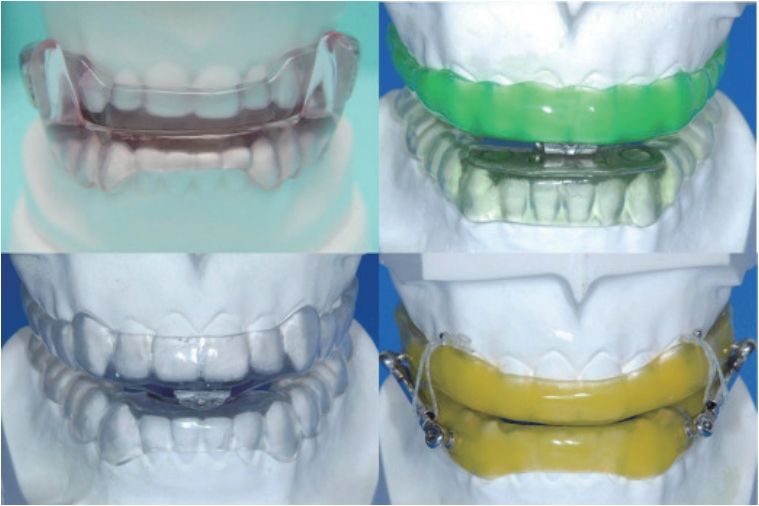
MADs, as the name suggests, function by moving the mandible in an anteroinferior direction. These appliances are further divided into custom-made and noncustom (or so-called ŌĆ£boil and biteŌĆØ) devices. The custom-made devices generally comprise the upper and lower portions and are positioned over the dentition in a single-piece (monoblock) (Fig. 1) [6] or two-piece (biblock) (Fig. 2) [6] according to the patientŌĆÖs preexamined bite registration [7]. While monoblock operates in a predetermined level of mandible advancement, biblock detaches the upper and lower splints to achieve the desirable degree of mandibular protrusion. Noncustom MADs, which are usually in a premade state, are usually composed of materials that are malleable on warm water exposure. Biting down aids in fitting the device, hence the name ŌĆ£boil and bite.ŌĆØ
TRDs (Fig. 3) [6] draw the tongue forward in a position without regard to the mandible. Since these are composed of a mouthpiece covering the upper and lower arches, TRDs can be recommended for patients with insufficient dentition. Though studies show that MADs and TRDs have similar effects in apnea reduction, more than 90% of patients preferred the MADs due to better compliance [8].
Palatal lift appliances elevate the soft palate through an adjustable acrylic button, thus preventing the tissue from vibrating during slumber. While this helps in reducing snoring for OSA patients [9], most of them have difficulty maintaining the appliances for an extended period. Consequently, palatal lift appliance use in OSA is becoming limited.
MECHANISM OF ORAL APPLIANCE
Direct Anatomical Action
By advancing the mandible in an anteroinferior direction, first, oral appliances draw the tongue forward by tracting the genioglossus [10]. This, in turn, leads to the advancement of the soft palate through tensioning the palatoglossus. Lastly, the pharyngeal soft tissues are positioned anteriorly as the mandibular ramus is placed at the front [11].
Counterintuitively, the combined actions of the involved tissues result in a greater increase of lateral dimension rather than anteroposterior dimension, which has been verified through multiple imaging studies (e.g., 3D videofluoroscopy, magnetic resonance imaging [MRI], computed tomography [CT], and drug-induced sleep nasendoscopy) [11-13]. The expansion of the lateral pharyngeal wall is presumably associated with a direct connection between the lateral pharyngeal wall and the ramus via the pterygomandibular raphe [11].
Indirect Anatomical Action
Rather than directly expanding the upper airway, oral appliances also work indirectly by preventing upper airway collapse. While the process involved remains vague, various studies attribute this to the tensioning of the postpalatal area as well as different mandibular positions [14,15]. Both of these processes intervene in intraluminal pressure during inspiration and upper airway muscular activity, resulting in a reduction of upper airway closing pressure [16].
Physiologic Action
Upper airway patency is also associated with neuromuscular reflex of the upper dilator muscles. As the mandible advanced with the implementation of oral appliances, masseter, lateral pterygoid, genioglossus, and geniohyoid muscles activation was demonstrated by surface electrodes [17-19]. However, a direct association between mandible advancement and shift in genioglossus activity is yet to be confirmed through studies with large sample sizes.
PATIENT SELECTION AND TREATMENT OUTCOME
Clinical Guideline
To date, the clinical guidelines provided by the American Academy of Sleep Medicine (AASM) and the American Academy of Dental Sleep Medicine (AADSM) in 2015 [3] and guidelines updated by the National Institute of Clinical Excellence (NICE) in 2021 [20] are used to determine the use of oral appliance in OSA.
Both guidelines state that oral appliance therapy is indicated for adult patients who cannot adhere to PAP treatment (due to device-related issues such as discomfort, mask leaks, skin irritation, and dry nose or mouth).
Additionally, they both assert that oral appliances should be prescribed by a dental expert, well acquainted with sleep medicine, to obtain informed consent, set the optimal titration level and design, and supervise both short- and long-term follow-up to establish ideal comfort and therapeutic effectiveness. A follow-up polysomnography (PSG) with the appliance in situ has also been emphasized in both guidelines to evaluate the changes in sleep physiology.
While both guidelines do not state a recommendation for a specific type of oral appliance (i.e., MAD, TRD, etc.), they both emphasize the use of ŌĆ£custom, titratable appliance over noncustom oral devices.ŌĆØ The NICE guidelines further warns against the use of noncustom oral appliances, as they provide impaired fit, for those with active periodontal disease or untreated dental decay, few or no teeth, and generalized tonic-clonic seizures.
Contraindication
To ensure a secure positioning and to prevent damage to the teeth and gingiva, MADs are contraindicated for patients with insufficient dentition, severe decay, loose teeth, gingivitis, or periodontitis. While dental implants, crowns, and caps are not contraindicated, their stability should be evaluated to avoid displacement and patients should be informed that possible future dental work such as braces would require a remake [21].
Since oral appliances, especially MADs, inevitably affect the mandible and temporomandibular joint (TMJ), they are contraindicated for patients with TMJ disease, inadequate mandibular protrusion, and severe micrognathia [21]. Oral appliance is relatively contraindicated for patients with nasal obstruction, large tonsils, elongated palate, and uvula as they can reduce efficacy [22].
Device Design
Various features should be considered prior to oral appliance use as they directly affect patient compliance and therapeutic efficacy and may have adverse effects [23,24].
Customization
While both custom and noncustom oral appliances reduce apnea-hypopnea index (AHI) in patients with OSA, a meta-analysis conducted by the AASM and AADSM revealed statistically significant differences in AHI reduction, minimum SaO2, and Epworth-sleepiness scale improvements in favor of customized oral appliances [3].
This may be explained by the fact that customization to a patientŌĆÖs dentition ensures greater retention, which is defined as the ŌĆ£resistance of the appliance to vertical movement away from the tissuesŌĆØ [25] and ŌĆ£that quality inherent in the oral appliance acting to resist the forces of dislodgement along the path of insertionŌĆØ [26]. A randomized controlled crossover trial by Vanderveken et al. [27] revealed that greater compliance in patients with customized oral appliance therapy was associated with better retention.
Mandibular titration
A meta-analysis by the AASM and AADSM shows comparable results in AHI reduction, SaO2 improvements, and reduction in Epworth-sleepiness scale in both customized titratable and customized nontitratable oral appliances [3]. However, titration is essential as the differences in severity vary in individuals with OSA and the optimal amount of degree of mandibular advancement cannot be predicted [28].
While there is no standardized titration procedure [29], advancing the mandible in measured increments over time and evaluating both the patientŌĆÖs symptoms and overnight oximetry is thought to maximize treatment outcome [30]. Though this process does not provide immediate short-term efficacy, it provides time for the patient to adjust, which decreases the possibility of side effects and increases patient compliance in the long term [28].
Vertical dimension of mouth opening
The degree of vertical mouth opening is an important factor in tolerance and efficacy [31]. During slumber, the muscles relax, which results in the rotation of the mandible in a posteroinferior direction. The subsequent decrease in tongue space and upper airway narrowing negatively affects treatment efficacy [32]. Therefore, the vertical degree of mouth opening must be kept at a minimum [33].
Predicting and Improving Treatment Response
Since oral appliance therapy does not invariably result in success [34], establishing methods to aid in selecting patients who will show response is clinically important. The absence of consensus on treatment success among various studies hinders the comparison of results among various studies [29]. Therefore, a valid clinical method to differentiate patients showing response to treatment is yet to be established [35].
Anatomically, anterior tongue movement, shorter soft palate length, larger oropharyngeal airway space, smaller mandible, and a lower hyoid bone position are associated with better treatment outcomes [36,37]. Two recent systematic reviews [38,39], however, discovered that due to numerous study design failures, these cephalometric indexes require additional studies to validate their significance.
Clinically, young female patients with smaller neck circumference, lower body mass index (BMI), and lower prevalence of cardiovascular disease are expected to achieve better outcomes [37,40,41].
Psychological and social factors (e.g., mood and bed partner satisfaction) may influence treatment success by improving compliance with oral appliances [42,43]. On the contrary, a personality type with negative affectivity (i.e., type D) negatively affects compliance with therapy [29].
Lower OSA severity as well as supine predominance is acknowledged as a factor for treatment success. PSG indexes associated with better treatment outcomes include lower loop gain and higher oxygen saturation [43-46]. Thus, these indexes should be considered by sleep clinicians when prescribing treatment for OSA patients.
Different methods, such as upper airway visualization and behavior with or without mandibular advancement simulation, have been proposed to reliably predict treatment outcomes. Drug-induced sleep endoscopy (DISE) is often used to assess the magnitude and location of pharyngeal collapse [47].
Patients displaying greater pharyngeal patency during mandible advancement on DISE showed good sensitivity for treatment success and its potential limitation can be supplemented by awake nasendoscopy through the Mueller maneuver, with mandibular advancement [48]. Aside from endoscopic examination, MRI and CT scans, through the construction of the patientŌĆÖs upper airway geometries, can simulate airflow changes with mandibular advancement; consequently, predicting treatment outcomes [49,50].
Awake examination of flow-volume loops, along with phrenic nerve stimulation was applied to determine the association between the collapse of the airway region and treatment success [51-53]. Other airway evaluations during awaking have established a negative correlation between higher nasal resistance and treatment failure [22]. Lastly, higher continuous positive airway pressure (CPAP) requirement (> 10.5 cm H2O) for patients with a history of CPAP use was associated with treatment failure [54].
ADVERSE EFFECTS AND THEIR MANAGEMENT
Minor, transient complications from oral appliance usually occur during the first week. While these generally resolve with increased use, other long-term side effects escalate over time and may become irreversible. Therefore, it is imperative to inform every patient about the potential adverse effects before treatment commences as they may affect long-term compliance.
Dental Effects
As oral appliances exude pressure on the teeth, pain in the dentition is reported in approximately 60% of patients [55]. To alleviate the pressure, the appliance should be grated lest the teeth loosen or fall off. If the pain continues, cessation of appliance use should be considered [21]. In cases of patients with previous dental cap or crown history, the prosthesis may come off. However, a primary repair from a skilled dentist is usually sufficient.
Occlusion, the contact between the upper and lower dentition, becomes altered as the oral appliance advances the mandible during sleep. Short- and long-term occlusion changes are reported in approximately 41% of patients [21]. Initially, patients experience an unusual sense of pain during mastication, predominantly on awakening. If the anterior positioning of the mandible persists, a permanent shift to class III malocclusion may occur. To prevent this, a premade morning aligner, in which the initial normal bite of the patient is imprinted, is used to adjust the mandible into the original position [56].
That said, a shift in occlusion can gradually occur with long-term oral application use. This is mostly associated with the extent of the vertical and horizontal overlap of the maxillary central incisors over the mandibular central incisors (i.e., overbite and overjet, respectively). A meta-analysis by Araie et al. [57], reported a 0.99-mm and 1.0-mm decrease in overjet and overbite, respectively, which contribute to the increase in the inclination of mandibular incisors. These changes become pronounced after 6 months of use [58] and the degree of change is correlated with the duration of treatment [59]. While the change in occlusion typically goes unnoticed in the majority of patients, a routine dental assessment is crucial as the change in occlusion and the subsequent difficulty in mastication is difficult to treat. Cessation of appliance use and use of orthodontics and surgical retrusion of the mandible for treatment may be considered [56].
Oral Cavity Effects
At the first and second week of application, 60% of patients complained of increased drooling and salivation [27], which usually subside as the patients acclimate. The patients are instructed to use a towel over the pillow to prevent staining of beddings [56]. On the other hand, desiccation of the oral cavity is reported in 86% of patients as a minor long-term side effect [55].
Due to the pressure applied to the gingiva and the oral mucosa at the early stage of use, transient irritation and ulceration occur. Tongue ulceration occurs in 8% of patients as the result of inadequate space provided and protrusion against the appliance [60,61]. The increase in space caused by slow advancement of the mandible, usually resolves the issue.
Musculoskeletal Effects
Due to the dentition change, caused by long-term oral appliance use, lower and anterior facial height increase on cephalometry is reported in patients with oral appliance use longer than 2 years [59,62]. However, as a meta-analysis reports no significant skeletal changes, the question of craniofacial change as an adverse effect is debatable [57].
Dysfunction in TMJ is relatively common after oral appliance use, especially for those with underlying joint disorders. While TMJ pain is reported in roughly 37% of patients [63], it generally subsides after 6 to 12 months [64,65]. TMJ pain may be caused by the sudden or excessive protrusion of an oral appliance. The usual instruction is to generally advance the device 0.5 mm two times a week. Should this result in TMJ pain, retreating a few millimeters puts less stress on the joint. Aside from directly manipulating the device, conservative measures such as analgesics and warm compression may be used as well [56].
COMPARISON OF ORAL APPLIANCES TO OTHER TREATMENTS
Continuous Positive Airway Pressure
While the indication for the use of oral appliances for OSA patients is expanding with consecutive iterations of AASM guidelines, the evidence associating oral appliance efficacy with disease severity is paltry. As such, CPAP remains the fundamental treatment of choice for OSA and the most recent AASM guidelines do not delineate a specific disease severity for oral appliance [3,66-68].
PSG index
Arguably, the assessment of the overall success of OSA treatment should also encompass the patientsŌĆÖ quality of life. However, physiologic variables demonstrated by PSG are easier to acquire and relatively more objective. Therefore, most studies comparing the treatment success between CPAP and oral appliance therapy focused on AHI and oxygen saturations when measuring outcomes.
While the task force assigned by the AASM and AADSM acknowledge that oral appliance therapy significantly improves AHI and oxygen saturation, through meta-analysis, they concluded that CPAP is more effective than all the other types of oral appliance (i.e., custom, titratable; custom, non-titratable; noncustom, titratable; and noncustom, non-titratable) regarding improvements to AHI and oxygen saturation with a mean difference of 6.24 events/hr and 3.11%, respectively [3].
Quality of life
Aside from improving physiologic indices, the incidence of snoring, daytime somnolence, and the effect on daily function [4] must also be considered when selecting OSA treatment. The common parameters used for assessment were the Epworth-sleepiness scale, Medical Outcome Study Short Form-36, and Functional Outcomes of Sleep Questionnaire.
According to a meta-analysis conducted by the AASM and AADSM, oral appliance appears to be effective for primary snoring in the absence of OSA. However, the scant amount of evidence prevents an effective comparison of primary snoring between oral appliances and CPAP. Oral appliance and CPAP showed similar efficacy when it came to subjective sleepiness or the general quality of life. However, the quality of evidence on this statement is low and additional studies are required for further assessments [3].
Compliance
While both CPAP and oral appliance therapy improve OSA, they would be obsolete if the patient disregards treatment. Therefore, compliance and adherence to therapy, are essential factors for treatment success.
Overall, patients adhered to oral appliance treatment, as confirmed by the AASM and AADSM [3]. Other meta-analysis reported up to 1.1 more hours of use in favor of oral appliances [69]. While this may be an explanation for reports of similar health outcomes between CPAP and oral appliance therapy [70], the adherence to oral appliance was measured according to the patientŌĆÖs subjective report questions, and therefore the validity of the data included in the analysis cannot be ascertained.
Cardiovascular outcomes
Various randomized controlled trials have already demonstrated favorable effects on cardiovascular outcomes with regular use of CPAP [71,72]. While one nonconcurrent cohort reported a significantly lower cardiovascular mortality rate in both the CPAP and oral appliance groups, the difference between CPAP and oral appliance therapy was not significant [73]. Therefore, large-scale randomized control trials comparing the impact of CPAP and oral appliance therapy are requir
Surgery
The efficacy of oral appliance and uvulopalatopharyngoplasty where pharyngeal tissues including uvula, soft palate, and palatine tonsils are removed, was compared in a randomized study. After 4 years of follow-up, oral appliance displayed a statistically significant difference in AHI improvement (5 events/hr) [74]. While the outcome is worthy of note, the oral appliance therapy compliance rate decreased to 62% in the oral appliance group at the end of the 4-year follow-up, which may have affected the results.
Further Research and Limitations
While oral appliances are being increasingly recognized as a safe treatment of choice for a selected group of OSA patients, a myriad of questions remain, which could be answered by future studies. To define the mechanism of oral appliances, additional studies are required to verify the relation between oral appliances and elements such as pharyngeal airway patency and genioglossus muscle [75]. While numerous publications discuss the predictive factors for the success of oral appliance therapy, the inherent flaw in the methodology yielded inconsistent results. As such, future research should focus on redefining the predictive factors of oral appliances and making them readily accessible to clinicians [3]. To individualize the best degree of mandible advancement, standardized titration procedures should be discussed in future studies. For the long-term effectiveness of oral appliance therapy, patients should be sufficiently informed about the potential side effects as well as ways to mitigate them [3]. To adequately compare the treatment efficacy between CPAP and oral appliances, future studies should adopt objective ways to monitor oral appliance adherence and use only custom-made titratable oral appliances.
CONCLUSION
While CPAP remains the mainstay of OSA treatment, a higher rate of adherence and similar health outcomes make oral appliances an appealing option, especially for those who are unable to sustain CPAP therapy. Organizing ways of selecting patients who can achieve optimal treatment outcomes with oral appliances as well as constructing customized devices capable of individual titration and monitoring, will improve the efficacy of oral appliances in the treatment of OSA.