AbstractBackground and ObjectiveMany studies have supported the potential effect of anti-inflammatory medications treatment for childhood obstructive sleep apnea (OSA). This study aimed to evaluate the outcomes of intranasal corticosteroid treatment in children with OSA symptoms and adenoid hypertrophy (AH), and to identify factors associated with the treatment outcome.
MethodsThis retrospective study included children aged 1–15 years who snored, had sleep-related symptoms, and received intranasal corticosteroid treatment for 4–16 weeks. Treatment outcome was considered a success if all symptoms were completely resolved or if the child still snored without sleep-related symptoms; treatment was considered a failure if the child still snored and had concomitant sleep-related symptoms.
ResultsAmong the 328 eligible children, 109 (33.2%) experienced treatment failure. The age of the children was significantly higher in the failure group than in the success group (median [interquartile range], 5.5 [3.4, 8.4] vs. 4.2 [3.2, 6.3]; p = 0.004). The percentage of children with allergic rhinitis (AR) symptoms at follow-up visit was also significantly higher in the failure group than in the success group (44% vs. 27.9% with mild AR and 9.2% vs. 4.1% with moderate-to-severe AR, p < 0.001). Multiple logistic regression revealed a significant association between treatment failure and older age (odds ratio [95% confidence interval], 1.13 [1.04–1.23]) and presence of mild AR (2.29 [1.39–3.77]) and moderate-to-severe AR (3.17 [1.20–8.39]).
INTRODUCTIONObstructive sleep apnea (OSA) is a sleep disorder characterized by episodic partial or complete upper airway obstruction during sleep. The prevalence of childhood OSA is varied, but most studies report a prevalence between 1% and 4% [1]. The predominant cause of OSA in children is hypertrophy of the tonsils and adenoids. Although polysomnography is the gold standard for OSA diagnosis, the disease is usually suspected on the basis of clinical symptoms reported by caregivers. Snoring is the hallmark symptom of OSA, and if it occurs in conjunction with airway obstructive symptoms or symptoms of morbidity, the possibility of OSA should be considered [2].
A previous study found that the adenoid tissues of children with OSA have a high abundance of glucocorticoid receptors [3]. Another in vitro study in children with OSA, wherein whole-tissue cell cultures of the adenoids and tonsils were incubated with corticosteroids, found a reduction in cellular proliferation and the production of pro-inflammatory cytokines in a tonsils and adenoids [4]. These findings support the potential effect of intranasal corticosteroids in reducing adenoid hypertrophy (AH). Several clinical studies have provided evidence for the efficacy of intranasal corticosteroid treatment on the basis of improvements in polysomnography findings [5-8], and reduced adenoid size [7]. Additionally, the OSA-18 survey score has been used as an outcome measure, with a decrease in the total score after intranasal corticosteroid treatment indicating a good outcome [9].
Although studies have indicated the effectiveness of intranasal corticosteroid treatment, in clinical practice, we encounter children who do not respond to this treatment. Furthermore, few studies have determined the factors associated with intranasal corticosteroid treatment outcome. Therefore, we conducted this study to evaluate the outcome of short-term intranasal corticosteroid treatment in children with OSA symptoms and AH and examined the associated factors.
METHODSStudy Design and PopulationThis retrospective study was conducted at the pediatric outpatient clinic of Songklanagarind Hospital, Prince of Songkla University in Southern Thailand. Patient information was collected from the hospital records by using the International Classification of Diseases, Tenth Revision codes. Children aged 1–15 years who attended the clinic between 2014 and 2018 with one of the following diagnoses were recruited for initial review: J352 (hypertrophy of adenoids), J353 (hypertrophy of tonsils with hypertrophy of adenoids), G473 (sleep apnea), G479 (sleep disturbance), and R065 (mouth breathing). Based on the initial review, children who met all the following criteria were enrolled as the study participants: 1) snoring for 3 nights or more per week for at least 4 weeks; 2) having at least one of the following nine sleep-related symptoms: difficulty in breathing, apnea, restless sleep or frequent awakening, sleeping in the sitting position, mouth breathing, choking, enuresis, morning headache, and daytime sleepiness; 3) AH defined using an adenoidal-nasopharyngeal (AN) ratio of more than 0.5 measured on lateral neck radiographic images; and 4) received intranasal corticosteroid treatment, with good compliance (medication use more than 90%), for 4–16 weeks before the first follow-up visit. Children were excluded from the study if they met any of the following criteria: used intranasal or systemic corticosteroids within 4 weeks before the OSA diagnosis; previous tonsil or adenoid surgery; and craniofacial, neuromuscular, or genetic disorders.
Ethical approval for study protocol was obtained from the Ethics Committee of Faculty of Medicine, Prince of Songkla University, Thailand (aproval no. REC 63-147-1-1).
Data CollectionPatient information was collected from medical records at the initial visit and the first follow-up visit. Data collected at the initial visit included age; sex; weight; height; tonsil size; presence of upper respiratory tract infection symptoms, allergic rhinitis (AR), or asthma; AN ratio; and types of intranasal corticosteroid and concomitant medications including montelukast, antihistamines, antibiotics, and asthma controllers. Data collected at the follow-up visit included the duration of intranasal corticosteroid use; presence of upper respiratory tract infection symptoms, AR, or asthma; and allergy skin test results.
Treatment OutcomeAt the follow-up visit, snoring and the following sleep-related symptoms were assessed: difficulty in breathing, apnea, restless sleep or frequent awakening, sleeping in the sitting position, mouth breathing, choking, enuresis, morning headache, and daytime sleepiness. The response choices were presence or absence of each symptom.
The treatment outcomes of all participants were evaluated and classified into four categories: 1) all symptoms completely resolved, 2) all sleep-related symptoms resolved but still snoring less than 3 nights per week, 3) some improvement but still snoring and having sleep-related symptoms, and 4) none of the symptoms improved. The first two categories indicated treatment success and the remaining two categories indicated treatment failure.
Definition and Measurement MethodsAR was considered if the child had rhinitis symptoms (sneezing, a runny or blocked nose, and nasal itching) and was diagnosed as having AR by a physician. AR severity was classified according to the Allergic Rhinitis and Its Impact on Asthma guideline [10]. Asthma was considered if the child had ever wheezed and was diagnosed as having asthma by a physician. Adenoid size was measured on lateral neck radiographic images according to a technique described by Fujioka et al. [11]. AH was considered if the AN ratio was more than 0.5. Tonsillar hypertrophy was considered if the child had a tonsil size of grade 2 or more. Obesity and morbid obesity were defined using a weight by height percentage of 140%–199% and > 200%, respectively.
Statistical AnalysisStatistical analysis was performed using R software 4.1.1 and epicalc package 3.5.2.0. (R Foundation for Statistical Computing, Vienna, Austria). The differences in patient characteristics in the treatment success and failure groups were compared using the chi-square test or Fisher’s exact test for categorical data. As the continuous variable (age and weight for height) are not normally distributed, then the Wilcoxon rank-sum test was used for the analysis. A further analysis was performed to examine the factors associated with treatment failure using multiple logistic regression. Statistical significance was set at p < 0.05.
RESULTSCharacteristics of the PatientsA total of 1210 patient records were initially reviewed and 328 eligible patients were included in the study (Fig. 1). The median age (interquartile range [IQR]) of the participants was 4.5 (3.2, 6.9) years. Among the patients, 45 children (13.7%) had obesity and 6 (1.8%) had morbid obesity. Enlarged tonsils, AR, and asthma were found in 89.6%, 38.4%, and 7.9% of patients, respectively. The children’s age was significantly lower with increasing AN ratio: median (IQR) age of 7.6 (4.5, 10.3), 5.4 (3.9, 8.1), 4.5 (3.3, 6.6), and 3.5 (2.5, 5.2) years for the AN ratio of 0.6, 0.7, 0.8, and 0.9, respectively (p < 0.001) (Fig. 2).
The most commonly prescribed intranasal corticosteroid was fluticasone furoate (88.4%). Changes in AR symptoms were found at the follow-up visit. The percentage of children with moderate-to-severe AR decreased from 15.9% at the initial visit to 5.8% at the follow-up visit. Of 126 children with AR symptoms at the initial visit, 48 (38.1%) had undergone allergy skin tests, and 29 (60.4%) had a positive test result.
Treatment OutcomeOf the 328 children, 21 (6.4%) showed complete resolution of all symptoms, and 198 (60.4%) showed resolution of all sleep-related symptoms but still snored (primary snoring). These two groups (219/328, 66.8%) were categorized as having treatment success. Of the remaining children, 70 (21.3%) had some improvement but still snored and had sleep-related symptoms, and 39 (11.9%) showed no improvement of symptoms. These two groups (109/328, 33.2%) were categorized as having treatment failure.
Comparison of the Treatment Success and Failure GroupsVarious characteristics that were considered as potential factors associated with treatment outcomes were examined (Tables 1 and 2). No significant difference was observed between the success and failure groups in term of sex, weight for height percentage, tonsil size, AN ratio, and presence of upper respiratory tract infection symptoms and asthma. Regarding the treatment, we found that the types and duration of intranasal corticosteroids used as well as concomitant medications were not significantly different between the groups. The variables that differed significantly between the treatment success and failure groups were age and the presence of AR symptoms at the follow-up visit. The median age of children was significantly higher in the failure group than in the success group (5.5 [3.4, 8.4] vs. 4.2 [3.2, 6.3], p < 0.01). The rate of treatment failure according to age groups is shown in Fig. 3. We found that children aged 9–13 years had the highest failure rate, which ranged from 60.9% to 64.3%. We also found that the percentage of children with persistent AR symptoms at the follow-up visit was significantly higher in the failure group than in the success group (44% vs. 27.9% with mild AR and 9.2% vs. 4.1% with moderate-to-severe AR, p < 0.01).
Multiple logistic regression was applied to evaluate the contribution of age and AR symptoms to treatment failure. A significant association was observed between treatment failure and older age (odds ratio [95% confidence interval], 1.13 [1.04–1.23]) and presence of mild AR (2.29 [1.39–3.77] and moderate-to-severe AR (3.17 [1.20–8.39]) (Table 3).
Management of Children Who Did Not Respond to an Intranasal CorticosteroidChildren who did not respond to an intranasal corticosteroid had further investigations. Overnight pulse oximetry was performed in 93 children and 27 were found 3 or more clusters of desaturation which indicated a positive test. Polysomnography was performed in 9 children and 7 were found abnormal. Adenotonsillectomy was performed in 93 children.
DISCUSSIONThis study among children aged 1–15 years who had OSA symptoms and AH showed that the overall rate of intranasal corticosteroid treatment success was 66.8%. Although the differences in study methodology between our study and other studies made comparisons difficult, our treatment results were similar to those of a study on 40 children aged 2–11 years with sleep-disordered breathing and AH; this study found that the mean OSA-18 score improved in 65.8% of children treated using mometasone furoate for 4 weeks [9]. The effect of fluticasone propionate nasal spray versus placebo in 25 children with OSA was investigated and found that 69% of children showed an improvement in their symptom score [5]. Another study on 62 children with mild OSA found normalization of polysomnography sleep measures in 54.1% of children after 6 weeks of treatment with budesonide nasal spray [7].
We identified two factors that were independently associated with treatment failure. The first factor was older age, which on average, increased the odds of having treatment failure by 1.13-fold for every point increase in age in years. No previous study has indicated such an association between a child’s age and response to intranasal corticosteroid treatment. Although we could not explain exactly why younger children with OSA and enlarged adenoids responded to intranasal corticosteroid treatment better than the older children did, we present three possible explanations. First, adenoid size could be the factor affecting the treatment outcome. We found that patients with a high AN ratio were younger children, and hence, the reduction in the size of the adenoids after intranasal corticosteroid treatment could be more clinically significant in these children than in older children with smaller adenoids. Second, the action of topical corticosteroids depends on the presence of glucocorticoid receptors in the tissue, and hence, the limited number of receptors in the adenoid tissues of older children potentially affected the treatment outcome. Third, the failure of intranasal corticosteroid treatment in older children may reflect that an enlarged adenoid might not be the main cause of OSA. Further studies are warranted to explore and confirm these possibilities.
The second factor associated with treatment failure in this study was the presence of AR symptoms at follow-up. We found that the prevalence of AR in our patients with OSA was 38.4%, which was higher than that in the general population in Thailand, with prevalences of 15% and 17.5% in children aged 6–7 years and 13–14 years, respectively [12]. A meta-analysis of studies on children with OSA found that the prevalence of AR was 45.2%, which was slightly higher than that of our study [13]. AR results in nasal obstruction because of mucosal edema and secretions. The negative pressure generated during inspiration in the nasal airway may predispose children to partial or complete upper airway obstruction during sleep.
Intranasal corticosteroids are highly effective for treating patients with AR. In our study, most children with AR showed a clinical improvement after intranasal corticosteroid treatment. However, a few children failed to respond and still showed AR symptoms. We found that the odds of having treatment failure (persistent OSA symptoms) were high in these children. A previous study of children with OSA who were treated with intranasal corticosteroids found that AR did not affect the treatment outcome [9]. The reason for the difference in the findings of the two studies is probably due to the difference in the AR definition used. Our study used presence of AR symptoms, whereas the previous study used a history of AR, regardless of symptoms. Our study results suggest that the level of control of AR, not just an allergic history, is an important determinant of the OSA treatment outcome.
For the management of patients with AR, several modalities should be incorporated with intranasal corticosteroids, e.g., oral and topical antihistamine, decongestants, and immunotherapy, which are highly effective in some cases. Avoiding allergens or minimizing exposure to known allergens can prevent allergic symptoms. In our study, the allergy skin test was performed in only 38.1% of children, and this implied that the allergens had not been identified in many children. Identifying allergens is a key factor in treatment planning, which also includes allergen avoidance and immunotherapy. However, as 40% of the skin tests yielded negative results in these patients with clinically diagnosed AR, a potential explanation is that these children might be allergic to less common allergens not included in the standard test. Another possible explanation is that these children had non-AR; although the management of non-AR is slightly different, intranasal corticosteroid treatment is still the management of choice for this condition [14].
Studies have suggested a relationship between AR and AH. Allergy is a risk factor for AH [15,16], and 22%–46.4% of children with AR also have AH [17,18]. However, the role played by allergies and allergens in adenoidal diseases remains unclear. Allergic diseases cause an inflammatory state that influences adenoidal tissue as well [19]. Adenoidectomy is the recommended treatment for childhood OSA, but if these children with OSA have AR as a comorbidity, the treatment outcomes might be unsatisfactory because of the persistent local allergic inflammation in the nasal mucosa [18]. Therefore, medical anti-allergic treatment is important for improving both nasal and OSA symptoms.
A limitation of this study is that we used subjective symptoms as the mainstay for OSA diagnosis. We did not use a previously developed sleep questionnaire because of the possibility of it containing incomplete data owing to the nature of the retrospective analysis. Additionally, studies have indicated that such questionnaires have low sensitivity and specificity and are not an effective diagnostic tool for childhood OSA [20]; moreover, cutoff scores to determine the success or failure of treatment are not available for these questionnaires. Moreover, we could not classify OSA severity on the basis of symptoms alone. Previous studies on intranasal corticosteroid treatment limited their study populations to children with mild OSA alone.
ConclusionThis study among children with OSA symptoms and AH showed that the overall rate of intranasal corticosteroid treatment failure was 33.2% and the associated factors were older age and presence of AR symptoms. Therefore, we recommend more intensive treatment if children with OSA and AH have AR as a comorbidity. These findings also suggest the need for further studies to determine the cause of the high treatment failure rate in older children.
NOTESAvailability of Data and Material
The datasets generated or analyzed during the study are available from the corresponding author on reasonable request.
REFERENCES1. Lumeng JC, Chervin RD. Epidemiology of pediatric obstructive sleep apnea. Proc Am Thorac Soc 2008;5:242-52.
2. Muzumdar H, Arens R. Diagnostic issues in pediatric obstructive sleep apnea. Proc Am Thorac Soc 2008;5:263-73.
3. Goldbart AD, Veling MC, Goldman JL, Li RC, Brittian KR, Gozal D. Glucocorticoid receptor subunit expression in adenotonsillar tissue of children with obstructive sleep apnea. Pediatr Res 2005;57:232-6.
4. Kheirandish-Gozal L, Serpero LD, Dayyat E, Kim J, Goldman JL, Snow A, et al. Corticosteroids suppress in vitro tonsillar proliferation in children with obstructive sleep apnoea. Eur Respir J 2009;33:1077-84.
5. Brouillette RT, Manoukian JJ, Ducharme FM, Oudjhane K, Earle LG, Ladan S, et al. Efficacy of fluticasone nasal spray for pediatric obstructive sleep apnea. J Pediatr 2001;138:838-44.
6. Alexopoulos EI, Kaditis AG, Kalampouka E, Kostadima E, Angelopoulos NV, Mikraki V, et al. Nasal corticosteroids for children with snoring. Pediatr Pulmonol 2004;38:161-7.
7. Kheirandish-Gozal L, Gozal D. Intranasal budesonide treatment for children with mild obstructive sleep apnea syndrome. Pediatrics 2008;122:e149-55.
8. Chan CC, Au CT, Lam HS, Lee DL, Wing YK, Li AM. Intranasal corticosteroids for mild childhood obstructive sleep apnea--a randomized, placebo-controlled study. Sleep Med 2015;16:358-63.
9. Jung YG, Kim HY, Min JY, Dhong HJ, Chung SK. Role of intranasal topical steroid in pediatric sleep disordered breathing and influence of allergy, sinusitis, and obesity on treatment outcome. Clin Exp Otorhinolaryngol 2011;4:27-32.
10. Brożek JL, Bousquet J, Agache I, Agarwal A, Bachert C, Bosnic-Anticevich S, et al. Allergic rhinitis and its impact on asthma (ARIA) guidelines-2016 revision. J Allergy Clin Immunol 2017;140:950-8.
11. Fujioka M, Young LW, Girdany BR. Radiographic evaluation of adenoidal size in children: adenoidal-nasopharyngeal ratio. AJR Am J Roentgenol 1979;133:401-4.
12. Chinratanapisit S, Suratannon N, Pacharn P, Sritipsukho P, Vichyanond P. Prevalence and risk factors of allergic rhinitis in children in Bangkok area. Asian Pac J Allergy Immunol 2019;37:232-9.
13. Cao Y, Wu S, Zhang L, Yang Y, Cao S, Li Q. Association of allergic rhinitis with obstructive sleep apnea: a meta-analysis. Medicine (Baltimore) 2018;97:e13783.
14. Poddighe D, Gelardi M, Licari A, Del Giudice MM, Marseglia GL. Non-allergic rhinitis in children: epidemiological aspects, pathological features, diagnostic methodology and clinical management. World J Methodol 2016;6:200-13.
15. Modrzynski M, Zawisza E. An analysis of the incidence of adenoid hypertrophy in allergic children. Int J Pediatr Otorhinolaryngol 2007;71:713-9.
16. Bozkurt G, Dizdar SK, Korkut AY, Coşkun BU. Adenoid vegetation in children with allergic rhinitis. Turk Arch Otorhinolaryngol 2015;53:168-72.
17. Said SA, Mchembe MD, Chalya PL, Rambau P, Gilyoma JM. Allergic rhinitis and its associated co-morbidities at Bugando Medical Centre in Northwestern Tanzania; A prospective review of 190 cases. BMC Ear Nose Throat Disord 2012;12:13.
18. Colavita L, Miraglia Del Giudice M, Stroscio G, Visalli C, Alterio T, Pidone C, et al. Allergic rhinitis and adenoid hypertrophy in children: is adenoidectomy always really useful? J Biol Regul Homeost Agents 2015;29(2 Suppl 1):58-63.
Fig. 1.Study design and flow diagram. INCS, intranasal corticosteroid; OSA, obstructive sleep apnea. 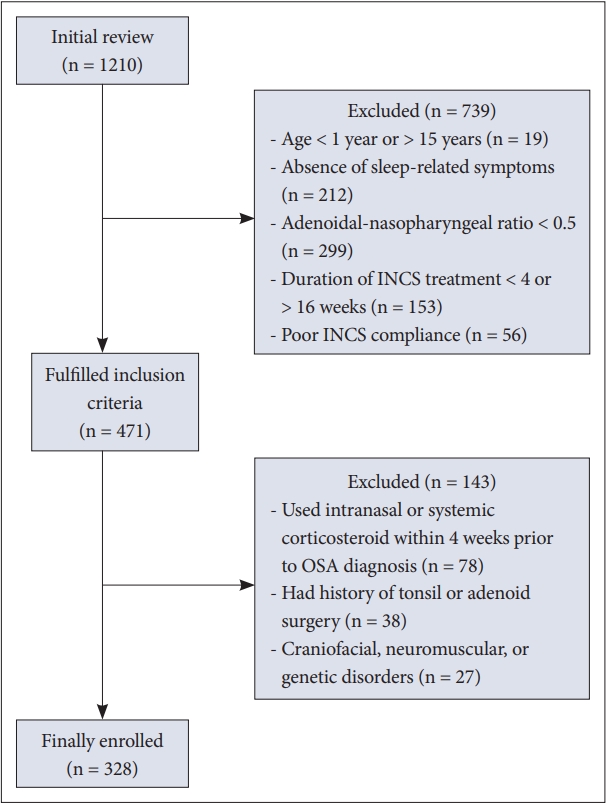
Table 1.Patient characteristics at the initial visit and outcomes of intranasal corticosteroid treatment Table 2.Patient characteristics at the follow-up visit and outcomes of intranasal corticosteroid treatment Table 3.Multiple logistic regression for factors associated with intranasal corticosteroid treatment failure |
|
||||||||||||||||||||||||||||||||||||||||||||