Prevalence, Risk Factors, and Impact of Excessive Daytime Sleepiness in an Elderly Korean Population
Article information
Abstract
Background and Objective
Excessive daytime sleepiness (EDS) is commonly reported by the elderly, but has not been well-described in older adults in Korea. This study aimed to estimate the prevalence and risk factors of EDS, and investigated the relationship between EDS and cognitive impairment in an older Korean population.
Methods
The cross-sectional and community-based study recruited 333 people ≥ 60 years of age. All participants underwent full-time nocturnal polysomnography and completed questionnaires assessing sociodemographic factors. EDS was defined as Epworth Sleepiness Scale score > 10. Cognitive functions were estimated using the Korean version of the Consortium to Establish a Registry for Alzheimer’s Disease neuropsychological assessment battery and subjective memory complaint questionnaire.
Results
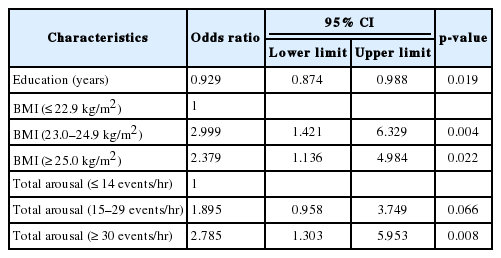
The overall prevalence of EDS was 18.9% with no significant difference between genders. In multivariate analysis using a multiple logistic regression model, obesity [odds ratio (OR) = 2.379, p = 0.022], educational attainment (OR = 0.929, p = 0.019) and total arousal (OR = 2.785, p = 0.008) were identified as the risk factors for EDS. Subjective memory impairment was related to EDS after controlling for confounding variables (p < 0.001).
Conclusions
Korean elderly have a high prevalence and risk factors of EDS, which has modest effects on cognitive functioning. EDS in older adults should be adequately addressed and treated, instead of being considered as a normal aging process.
INTRODUCTION
Excessive daytime sleepiness (EDS) is defined as the inability to stay awake and alert during major waking episodes of the day, resulting in unintended lapses into drowsiness or sleep, based on the second edition of International Classification of Sleep Disorders.1 EDS is one of the most disabling conditions caused by poor nocturnal sleep and is common in the elderly. Up to 30% of older people globally experience EDS.2 EDS can lead to poorer occupational and social functioning,3 and is strongly related to an increased risk of vehicular and occupational accidents.4 EDS also reduces cognitive functions in the elderly.5 Despite these adverse effects of EDS, the accurate representations of the burden of EDS in elderly Koreans are unclear. While several population-based studies of EDS in elderly people have been conducted in other countries,6–9 to our knowledge no study has evaluated EDS in older adults in Korea.
The causes of EDS are multifaceted, with various risk factors. A number of studies of older adults have examined contributory factors associated with EDS in the elderly. EDS often occurs because of severe sleep-disordered breathing, subjective poor sleep quality, increased rapid eye movement sleep, pain, and use of sedating medications.10 Risk factors for EDS are nighttime awakening, depression, snoring, non-use of sleeping pills, and limitations in mobility.11
Accumulating evidence suggests that sleep is pivotal for memory consolidation, maintaining attention and the ability to concentrate. Sleep seems to stabilize and enhance various types of memories.12 Sleep disturbance in elderly people deteriorates attention and concentration.13 Given that daytime sleepiness is strongly linked to poor nocturnal sleep, EDS could be related with cognitive dysfunctions. Several studies that addressed the relationship between EDS and cognitive impairment yielded inconsistent findings. EDS was independently associated with cognitive decline in older persons in one study,14 while other evidence prompted the suggestion that cognitive performance was not associated with the sleep-related daytime complaints.15
There have been few previous EDS studies in the elderly. Their findings were contentious and the studies were hampered by several limitations. Most were carried out in western populations,6–8,10,11,14 and involved self-administered questionnaires, without objective sleep measurements like polysomnography.6–8,11 In addition, only a few studies have investigated the relationship between EDS and cognitive dysfunctions. To overcome these limitations, we examined the prevalence of EDS and described associated lifestyle and sociodemographic factors of EDS in an elderly Korean population. This study also aimed to determine the relationship between EDS and cognitive decline in these Koreans.
METHODS
Study Sample
Data were obtained from the Korean Longitudinal Study on Cognitive Aging and Dementia, a population-based prospective cohort study of cognitive function with aging and dementia in the Korean geriatric population. Ten percent (n = 696) of 6959 adults ≥ 60 years of age residing in Jukjeon-1-dong, Sujigu, Yongin-si, South Korea, based on the residential roster were randomly selected. From November 2010 to January 2012, study research assistants telephoned and invited these subjects to visit Seoul National University Bundang Hospital (SNUBH). Among the 696 people, 466 received a clinical evaluation and 354 underwent nocturnal polysomnography (NPSG). Twenty subjects were excluded for short total sleep time (TST < 180; n = 6) and missing sociodemographic or clinical data (n = 15). Finally, 333 participants were included in the statistical analysis. This study was approved by the Institutional Review Board of SNUBH (B-1006/103-010). All participants or primary caregivers provided signed informed consent.
Sociodemographic Factors and Medical Information
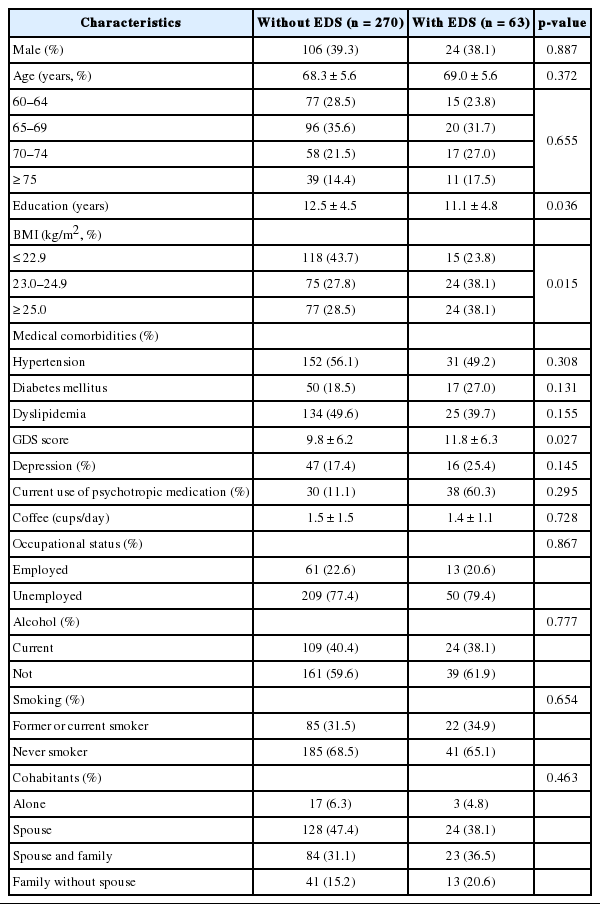
Self-administered questionnaires were used to collect information regarding sociodemographic factors including age, gender, body mass index (BMI, kg/m2), education level in years, alcohol consumption (current or non-current), caffeine consumption (cups per day), tobacco use (current smoker or never smoker), occupational status (employed or unemployed), and cohabitation status (alone, with spouse, with spouse and family, and with family but without spouse). To clarify the effects of obesity on the development of EDS, the participants were stratified into three BMI categories—≤ 22.9 kg/m2; normal, 23–24.9 kg/m2; overweight, and ≥ 25.0 kg/m2; obese—in accordance with the definition for obesity in Asian populations. History of medical condition was determined by the presence of hypertension, diabetes mellitus, and dyslipidemia based on questionnaires completed by participants or their primary care-givers. Enrolled subjects were also asked to report the current use (yes or no) of benzodiazepine or non-benzodiazepine sedatives. The geriatric depression scale (GDS) was used to screen for depression and to identify the severity of depressive symptoms. A score ≥ 16 was considered diagnostic for geriatric depression on the basis of the research on the validity and reliability of the Korean version of GDS.16
Measurements of Sleep
All participants underwent a full-time NPSG using a model N 7000 recording system (Embla, Reykjavik, Iceland) equipped with standard electrodes and sensors. Electromyography electrodes were also applied at the submentalis muscle and both anterior tibialis muscles to record nocturnal periodic limb movements. The epoch of the NPSG was scored every 30 seconds based on prior criteria.17
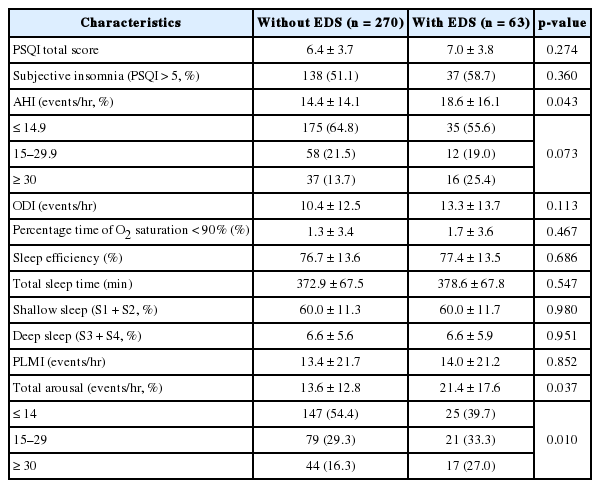
Apneas were defined as the complete cessation of airflow ≥ 10 seconds and hypopneas were defined as ≥ 50% reduction in airflow lasting more than 10 seconds, associated with either 4% oxygen desaturation or arousal due to ongoing respiratory effort. Apnea-hypopnea index (AHI) was calculated by the sum of apnea and hypopnea episodes per hour of sleep. According to widely accepted criteria for obstructive sleep apnea (OSA), study participants were distributed into three groups: healthy subjects (AHI ≤ 14.9), subjects with mild-to-moderate OSA (15 ≤ AHI ≤ 29.9) and subjects with severe OSA (30 ≤ AHI). The subjects were further subdivided into mild, moderate, and severe sleep fragmented groups (total arousal index, TAI; ≤ 14, 15 to 29, and ≥ 30, respectively). The following classifications were based on the criteria for OSA, since sleep-related respiratory events are closely related to arousals from sleep. Oxygen desaturation index (ODI) was the number of desaturation (≥ 4%) events per hour of sleep. Periodic limb movement index (PLMI) was computed as the number of the limb movements per hour of sleep. TAI was defined as the number of interruptions of sleep lasting 3 to 15 seconds per hour of sleep. Shallow sleep and deep sleep referred to the sum of time spent in stage 1–2 sleep and stage 3–4 sleep, respectively.
Diagnosis of EDS was made by means of the Epworth Sleepiness Scale (ESS), with a cut-off score exceeding 10. The ESS is a well-validated and widely-used subjective method to assess the levels of daytime sleepiness, with higher scores reflecting greater somnolence at daytime. It contains eight items evaluating the likeliness to fall asleep in eight different situations on a scale of 0–3. Subjective sleep quality was evaluated using the Pittsburg Sleep Quality Index (PSQI). The PSQI consists of 17 items with questions rated on a 4-point Likert scale ranging from 0 (very good) to 3 (very bad). The questionnaire indicates a global score based on seven components of sleep difficulty including subjective sleep quality, sleep latency, sleep duration, sleep efficiency, sleep disturbances, use of sleeping pills, and daytime dysfunction. All of these scores are summed to a global score, with scores > 5 indicating subjective insomnia.
Measurements of Cognitive Functions
The Korean version of the Consortium to Establish a Registry for Alzheimer’s Disease neuropsychological assessment battery was applied to evaluate cognitive functions. This battery includes Mini-Mental Status Examination, verbal fluency test, Boston naming test, word list memory test, word list recall test, word list recognition test, constructional praxis test, constructional recall test, and constructional recognition test. In addition to these tests, the study subjects were asked to perform Benton visual retention test A/C, trail-making test A/B, and Stroop color and word test. Subjective Memory Complaints Questionnaire (SMCQ) scores were also used to estimate the severity of subjective cognitive impairment among participants. The SMCQ consists of 14 items evaluating subjective memory complaints with higher scores indicating more severe memory dysfunctions.
Statistical Analyses
All statistical analyses were conducted on SPSS version 19.0 for Windows (SPSS Inc., Chicago, IL, USA). Characteristic differences between study subjects with and without EDS were analyzed using independent t-tests for continuous variables and chi-square tests for categorical variables. All the variables associated with the outcome in the univariate analysis were included in the multivariate model. Backward stepwise logistic regression model was used to identify risk factors for EDS after controlling for potential confounders. In addition, analysis of covariance (ANCOVA) adjusted by gender, age, educational level, GDS score, AHI, and TAI was adopted to determine the differences in cognitive functions between the two groups. All data are expressed as mean and standard deviation (SD) if not otherwise specified. Two-tailed p-values < 0.05 were considered statistically significant and the confidence interval was 95%.
RESULTS
Prevalence of Excessive Daytime Sleepiness
Among the 333 participants, 63 were diagnosed as EDS representing a prevalence of 18.9%. Overall, 24 men (18.5%) and 39 women (19.2%) reported EDS indicating a slight female predominance but no substantial difference was found between genders (p = 0.887). The mean age of the participants in this study was 68.43 years (SD = 5.59) with 39% males and 61% females and the mean BMI was 23.71 kg/m2 (SD = 2.72). The mean ESS scores were 6.67 (SD = 4.13), 13.22 (SD = 2.72) and 5.14 (SD = 2.25) in the total study population, EDS group and non-EDS group, respectively. After allocation into four groups on the basis of age (60 to 64 years, 65 to 69 years, 70 to 74 years, and ≥ 75 years), the rate of the presence of EDS exhibited a tendency to increase with age until the age of 74 years (16.3%, 17.2%, 22.7%, and 22.0%, respectively). However, there was no statistically significant difference across the age groups (p = 0.655).
Risk Factors of Excessive Daytime Sleepiness
Comparison data of sociodemographic and clinical characteristics of the study subjects with and without EDS using a univariate model are represented in Table 1. No substantial differences were observed between the two groups regarding gender, age, coffee and alcohol consumption, occupational status, tobacco use, cohabitants, medical comorbidities, presence of depression, and current use of medication. In contrast, pathological somnolence more prevalent in obese people (BMI ≥ 23.0 kg/m2) compared with normal-weight persons (BMI ≤ 22.9 kg/m2) (p = 0.015). Among subjects with EDS, a higher proportion reported having low education levels (p = 0.036) and more depressive symptoms (p = 0.027) than those without EDS.
Table 2 summarizes the sleep parameters of our study sample. Univariate analysis revealed significant associations between EDS and sleep factors including AHI (p = 0.043) and TAI (p = 0.037). EDS was correlated with sleep fragmentation (p = 0.010), but not sleep-disordered breathing (p = 0.073) after stratifying subjects into several groups based on the severity of symptoms. Individuals with and without EDS did not significantly differ with respect to the presence of insomnia and other polysomnographic findings such ODI, sleep efficiency, TST, percentage of sleep stages and PLMI.
The results of logistic regression analysis used to evaluate the association between EDS and potential risk factors are expressed in Table 3 with odds ratio and their 95% confidence intervals. Multivariate analysis revealed that education, obesity and total arousal remained significantly associated with EDS after adjusting for confounding effects (all p < 0.05).
Cognitive Impairment in Excessive Daytime Sleepiness
Table 4 indicates the comparison of cognitive function tests scores between older adults with and without EDS. After correcting for confounding factors using ANCOVA, significant differences were evident in SMCQ scores (p < 0.001) and constructional recall test scores (p = 0.017) across the two groups. EDS was associated with poor performance in both tests. No differences were found in mini-mental state examination, verbal fluency test, Boston naming test, word list memory test, word list recall test, word list recognition test, constructional praxis test, constructional recognition test, Benton visual retention test, trail-making test, and Stroop color and word test.
DISCUSSION
This population-based study indicates that EDS is common in Korean older adults (18.9% of community-dwelling elderly). Studies conducted in various countries have shown a wide variation in the prevalence of EDS ranging from 1.1% to 25%.6–9 These inconsistent findings might be caused by differences in methods and tools used to define EDS.
Several studies have reported gender differences regarding the presence of EDS, with positive associations between EDS and male gender in three studies,7,9,11 whereas two other studies reporting higher prevalence of EDS among females.8,18 In our study, a slightly higher rate of EDS in females was found, but individuals with and without EDS did not differ with respect to sex. Our finding is supported by a previous population-based study that demonstrated no association between the occurrence of EDS and gender.19 These dissimilarities could reflect methodological differences between studies. Furthermore, there has been no clear evidence for pathophysiological mechanisms underlying gender difference in EDS.
To identify risk factors for EDS in older adults, a cross-sectional study design was used to compare the presence of risk factors in subjects with and without EDS. Univariate analysis revealed that obesity, educational level, GDS score, AHI, and TAI could be associated with EDS. Multivariate analysis determined that only obesity, lower education, and frequent arousal as EDS risk factors.
An emerging literature has documented a link between obesity and excessive sleepiness. Obesity exacerbate sleep disordered breathing20,21 and sleep-related respiratory events might disturbed or shorten sleep, leading to fatigue and daytime sleepiness.22,23 Presently, there was a positive relationship between the severity of obesity and the development of EDS, whereas there was no significant difference between cases and control subjects in AHI, which serves as a predictor of sleep-related breathing disorders. Accumulating evidence indicates that obesity per se is strongly correlated with EDS independent of the presence of SDB.24,25 The underlying pathophysiological mechanisms of pathological somnolence associated with obesity are not clear. One possible explanation is that the mechanical effects of obesity may result in poor quality of sleep, such as sleep fragmentation and circadian shift.24 An alternative explanation is that EDS could be the consequence of elevated levels of inflammatory cytokines. Tumor necrosis factor-α and interleukin-6 levels are increased in obese people and those who exhibit EDS, while no increase was reported in normal-weight healthy controls.26 Furthermore, those with EDS are more likely to exercise less and gain more weight than healthy people. This reciprocal interaction between EDS and obesity may result in worsening of daytime somnolence in obese people, forming a vicious cycle.
Previous studies have suggested that sleep-related breathing disorders may result in daytime sleepiness.22,23 In contrast, the present data indicate that sleep-related respiratory problem is not an independent contributor to the risk for EDS in the geriatric population. Age-related changes in the cause of EDS could be responsible for less severe symptoms of sleepiness in elderly individuals with SDB. A recent survey observed an age-related reduction in the correlation between SDB and daytime sleepiness in males.27 Several previous studies reported age-related changes in structure and function of the upper airway28–30 and increased upper airway collapsibility during sleep in older age.31 These changes may lead to SDB with minimal sleep disturbance. For this reason, sleep-related breathing disorders could have a weak association with EDS in the elderly, as presently found.
The TAI was also found to be a significant risk factor for EDS. Total arousals comprise arousals associated with breathing difficulties or limb movements during sleep and unknown arousals. It is widely accepted that frequent nocturnal awaking might cause disruption of normal sleep structure, which is assumed to be responsible for EDS. Our results support previous indications that inadequate sleep could be one of the most important causes of EDS.32–34 In a recent study, entropy-based measures that were more accurate than traditional ones for quantifying sleep fragmentation reiterated the relationship between fragmented sleep and daytime sleepiness.35
Presently there was a substantial difference in the educational level between subjects with and without EDS. An explanation is that educational status might be related to daytime sleepiness through its associations with depression and anxiety. According to a large population-based cohort study, lower educational levels showed a robust association with both depression and anxiety.36 Another study also documented a significant inverse relationship between educational achievement and anxiety disorders.37 Also, several studies confirmed that depressed people are more likely to experience more fatigue and more severe daytime sleepiness.25,38 Although multivariate analysis revealed that depression may not be an independent risk factor for EDS in this study, higher GDS scores were evident in individuals with EDS. Furthermore, univariate analysis indicated that the severity of depressive symptoms could be a potential risk factor. In addition, undiagnosed anxiety symptoms could influence the subjective report of excessive sleepiness.
The current results demonstrate that a positive relationship between daytime sleepiness and self-reported memory decline after controlling for various confounding factors. In contrast, all the objective cognitive tests conducted in this study, except for the constructional recall test, did not differ significantly in subjects with and without EDS. Since the scores on the construction recall test reflect the ability to recall visual information, the finding could prompt the speculation that EDS has a detrimental effect on visual memory function. However, all the other cognitive tests scores including constructional praxis test and construction recognition test, which are also related to the registration and recall of visual memory, revealed no significant difference between subjects with and without EDS. Therefore, we cannot conclude that EDS is related to visual memory dysfunction and EDS may not be independently associated with the presence of objective cognitive deficits in the elderly.
There were some potential limitations. There was no objective assessment of the degree of daytime sleepiness. Multiple sleep latency test and maintenance of wakefulness test are gold standard tests to evaluate daytime sleepiness. Thus, further research incorporating these gold standards might be required to elucidate EDS more accurately. However, the ESS is a valid and widely-used tool for measuring daytime sleepiness and is cheaper, simpler, and less time-consuming. Perceived sleepiness based on the ESS score yielded moderate correlation with objective measures of sleepiness.39 Moreover, measures of objective sleepiness quantify sleep propensity during only a single day, while the ESS covers a longer period of time. Second, the cross-sectional study design was unable to determine the temporal relation between EDS and risk factors so that the direction of causality remains unknown. Cross-sectional methodology could not confirm causal relationship between EDS and cognitive dysfunctions either. Yet the aforementioned possible interpretations of our findings might be helpful to clarify causal linkage between EDS and its associated phenomena. Third, we should consider the first-night effect, which means disruption of usual sleep pattern due to unfamiliar environment of sleep laboratory. The best way to avoid this pitfall is performing NPSG twice for adaptation. But this methodology is rarely used in large population-based studies because of a substantial financial and time burden. Finally, we cannot exclude the effect of selection bias due to relatively low response rate because those who are concerned about their sleep were more willing to undergo clinical assessments including polysomnography in our study. This suggests that complaints of daytime sleepiness could be more frequent in our sample compared to the general population.
In conclusion, our study demonstrates that EDS is quite prevalent in the Korean elderly over 60 years of age. EDS is correlated with obesity, less-educated status, subjectively perceived daytime dysfunction and poor nocturnal sleep quality. Given that most of these risk factors are easily identifiable and also modifiable, clinicians should be aware of them to prevent debilitating and potentially dangerous health consequences of EDS. According to the results of our study, EDS also might engender subjective memory impairment. It is necessary to see if excessive sleepiness during the day is an early indicator for cognitive deficits in future investigations. Considering the high prevalence of EDS and marked impacts on the lives of Korean older adults, providing an optimal management of pathological somnolence to the old patients has a significant clinical implication. Further longitudinal and methodologically rigorous studies with the objective evaluation of sleepiness using the multiple sleep latency test and maintenance of wakefulness test are needed to confirm these findings and to extend our knowledge of the clinical aspects of EDS in the elderly.
Notes
Conflicts of Interest
The authors have no financial conflicts of interest.